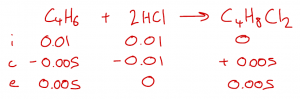Percentage yield is defined as the actual mass of product divided by the theoretical maximum mass of product, multiplied by 100%.
Equivalently, we can express this as the actual moles of product divided by the theoretical maximum moles of product, multiplied by 100%.
The theoretical maximum moles of product depends on the moles of the limiting reagent and the balanced symbol equation. However, the actual moles of product will also depend on the reaction (e.g., are there side reactions? is the reaction incomplete?) apparatus (e.g., is some product lost during transfer?), and the skill of the person doing the practical!
| Worked Example: Buta-1,3-diene reacts with HCl through electrophilic addition. HCl can be added either once or twice to each diene molecule forming a series of position isomers with the formulas C4H7Cl and C4H8Cl2. 0.540 g of C4H6 react with 50 cm3 of 0.2 mol dm-3 HCl. The mass of 2,3-dichlorobutane formed was 0.254 g. What is the % yield? We first need to calculate the amounts of each reactant: n C4H6 = 0.540 / 54 = 0.01 mol n HCl = 0.2 x 50 / 1000 = 0.01 mol Equation: C4H6 + 2 HCl –> C4H8Cl2 Using an ICE table, we can see that 0.01 mol of HCl reacts with 0.005 mol of C4H6 forming 0.005 mol of C4H8Cl2. This is the maximum theoretical moles of C4H8Cl2. The limiting reagent in this case is HCl because it is completely used up. The actual moles of C4H8Cl2 (Mr = 127) formed is 0.254 / 127 = 0.002 mol so the % yield is 0.002 / 0.005 x 100 = 40%. An obvious way to increase the % yield would be to ensure that HCl is in excess. This would ensure that any C4H7Cl (i.e., which has one remaining C=C) reacts further to form the dihalogenoalkane. |